Technology
If you have any questions or interests in our service
and business, please contact us.
Inquiry form here
Ecobody Technology
About Ecobody
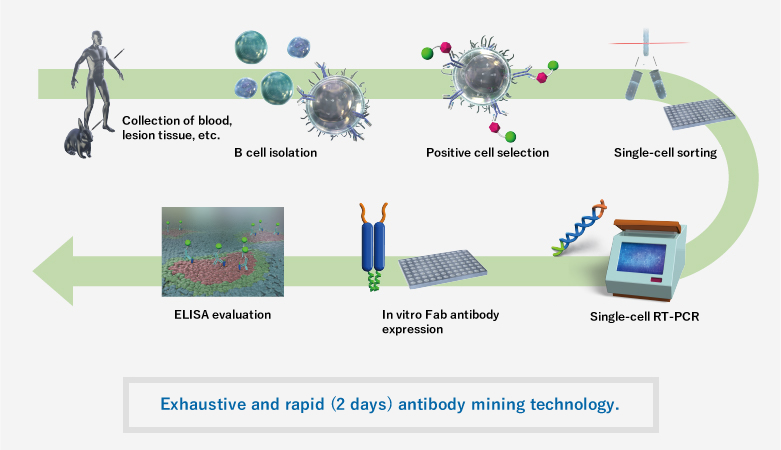
Benefits of “Ecobody” Technology

Related Patents
・Tagged antibody(Japan Patent No. 6744670)
・Protein Expression Method(Japan Patent No. 6681625; U.S. Patent Number: 10975376)
・PROTEIN EXPRESSION METHOD(European patent No.EP3312278)
※iBody has been granted an exclusive global license for these patents from Nagoya University.
Features of Antibody Mining Technologies
| Ecobody | Hybridoma | Phage Display | |
|---|---|---|---|
| Animals | Rabbit, Human | Mouse | Not used |
| Time | 2 days※ | A few months | 7 weeks |
| Culture | Unnecessary | Necessary | Necessary |
| Core Technology | Single-cell technology and in vitro antibody expression | Cell fusion and culture | Bacterial expression |
| Native Antibody Isolation | Possible | Possible | Impossible |
Rabbit Monoclonal Antibody

Achievements of monoclonal antibody mining from rabbits using “Ecobody” technology
Monoclonal antibody binding to a low-molecular-weight compound
Obtained monoclonal antibodies recognize a low-molecular-weight bioactive substance with high affinity and specificity
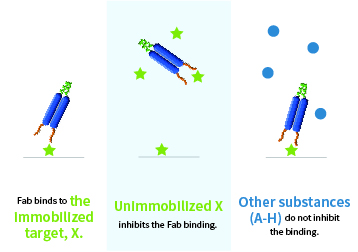
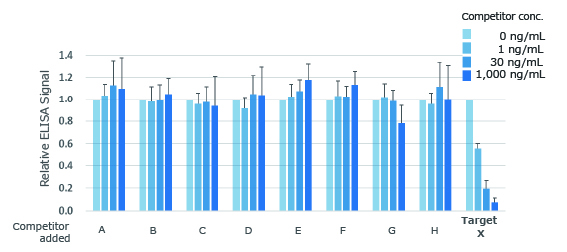
Unimmobilised target substance (X) inhibits the antibody binding, whereas other related substances (A-H) do not.
Monoclonal antibody detecting a post-translational modification
Obtained monoclonal antibodies specifically detect the protein phosphorylation.
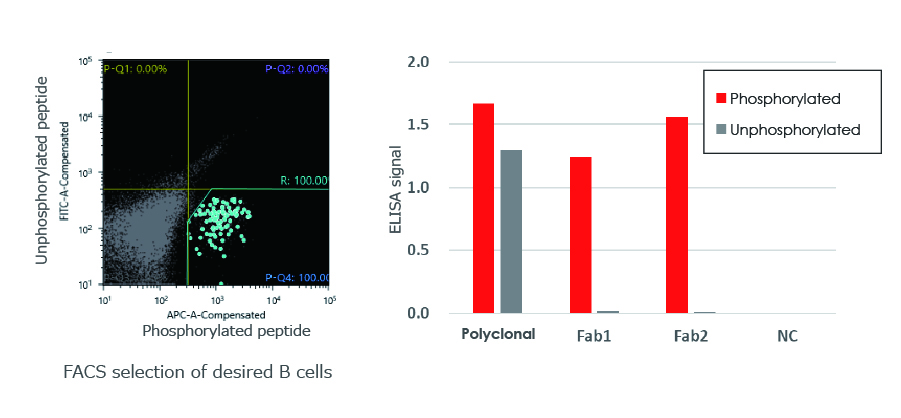
Antibodies obtained (Fab1, Fab2) selectively react with the target phosphorylated peptide.
Monoclonal antibody detecting slight amino acid sequence differences
Obtained monoclonal antibodies distinguish between slightly different amino acid sequences
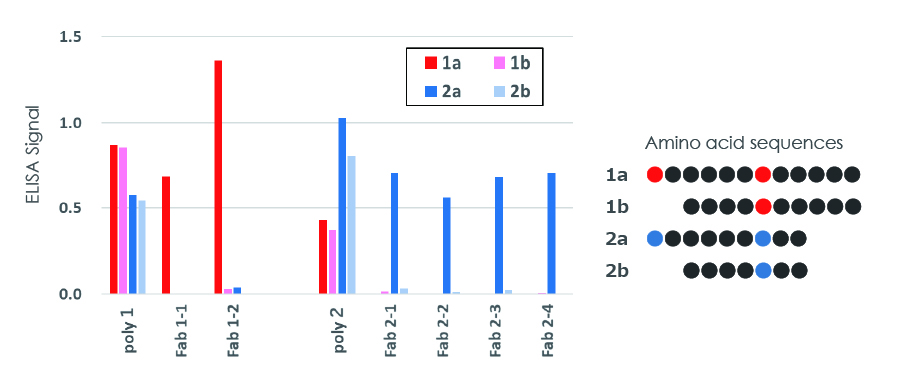
Rabbits immunized with antigens 1a and 2a produced polyclonal antibodies (poly1 and poly2, respectively) with low specificities. Monoclonal antibodies (Fab1-1-Fab2-4) obtained from these rabbits showed strong specificity for the target antigens.
Human Monoclonal Antibody

Achievements of monoclonal antibody isolating from humans using “Ecobody” technology
Monoclonal antibody recognizing a solid tumor
Target tumor-binding monoclonal antibodies were isolated from the cancer tissues of a solid tumor patients
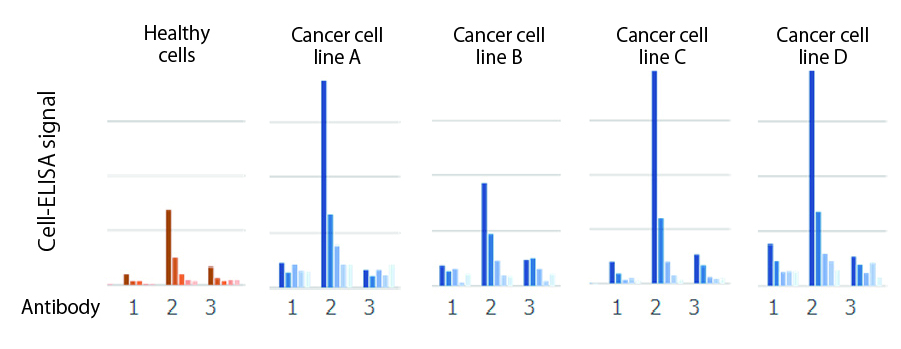
Obtained natural human antibody (#2) strongly binds to multiple lines of human cancer cells
Fluorescent immunostaining of xenograft cancer tissue of a model mouse
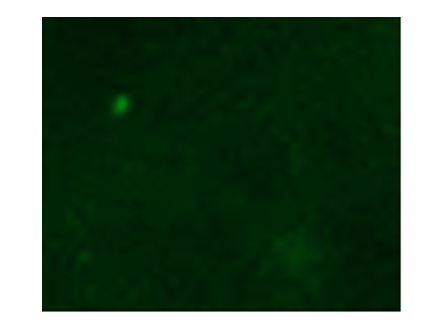
Control
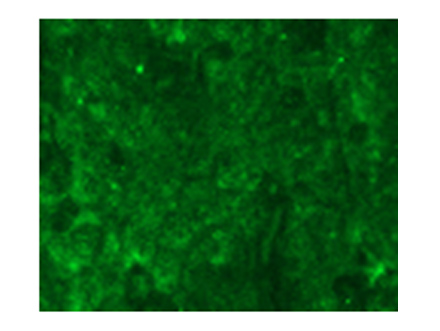
Antibody(#2)
Antibody obtained from a tumor patient is applicable for cancer tissue immunostaining
Monoclonal antibody against SARS-CoV-2
The monoclonal antibody (MO1) with neutralizing activity against diverse SARS-CoV-2 variants(D614G, Delta, BA.1, BA.1.1,BA.2, BA.2.75, and BA.5) was identified from blood samples of infected individuals.Neutralizing activity of the antibody MO1 against Omicron BA.5 variant
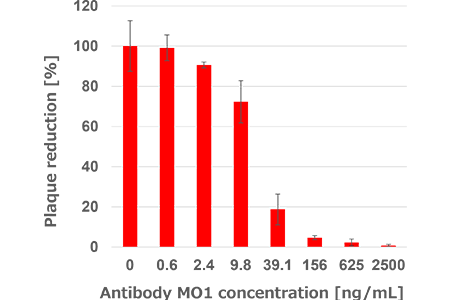
Antibody MO1 inhibited the viral proliferation in a concentration dependent manner.(The data was provided from Prof. Yasuko Mori of Kobe University)
Three-dimensional structure of the antibody MO1 binding to the target spike protein
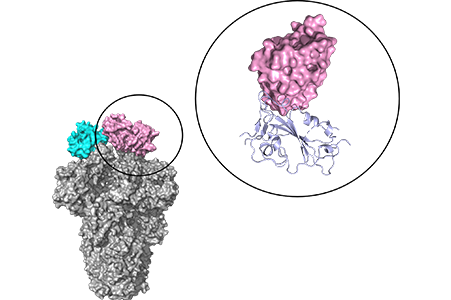
The antibody MO1 recognizes common sequence to various mutants(D614G, Delta, BA.1, BA.1.1, BA.2, BA.2.75, BA.5)MO1 bound to the purified target RBD proteins of BA.2 and BA.5 variants with the dissociation constants(Kd)of 3.3 nM and 11 nM, respectively.(Figure created from public information on PDB 8h3n)
References
- Rapid Generation of Monoclonal Antibodies from Single B Cells by Ecobody Technology. Antibodies, 7, 38 (2018)
- Ecobody technology: rapid monoclonal antibody screening method from single B cells using cell-free protein synthesis for antigen-binding fragment formation. Sci Rep, 7, 13979 (2017)
- N-terminal SKIK peptide tag markedly improves expression of difficult-to-express proteins in Escherichia coli and Saccharomyces cerevisiae. J Biosci Bioeng. 123, 540 (2017)
- ‘Zipbody’ leucine zipper-fused Fab in E. coli in vitro and in vivo expression systems. Protein Eng Des Sel. 29, 149 (2016)


